Depression is not simply sadness. It is a complex biological condition rooted deeply in brain chemistry, neural circuitry, and structural changes that develop over time. When people explore options like Ketamine Treatment for Depression, they are often curious about one key question: what exactly happens in the brain, and how does it change over weeks and months?
To understand this, we need to look beneath symptoms and into the microscopic world of neurons, neurotransmitters, and synapses. Ketamine interacts with the brain in a way that is fundamentally different from traditional antidepressants. Its effects unfold in stages, from rapid chemical shifts to longer term structural adaptations.
Understanding Depression at the Neurochemical Level
The Role of Neurotransmitters in Mood Regulation
Your brain communicates through chemicals called neurotransmitters. Think of them as messengers passing notes between billions of nerve cells. Serotonin, dopamine, norepinephrine, and glutamate are some of the most important players in mood regulation.
In depression, this communication network becomes disrupted. Signals weaken. Connections shrink. Entire circuits involved in motivation, pleasure, and emotional regulation may slow down.
But depression is not just about having too little serotonin. It also involves inflammation, stress hormones like cortisol, and structural changes in key brain regions such as the prefrontal cortex and hippocampus.
Why Traditional Antidepressants Target Serotonin
Most antidepressants, such as SSRIs, increase serotonin levels gradually. Over weeks, this adjustment helps rebalance signaling. However, these medications often take four to eight weeks to show significant improvement.
Why so slow? Because the brain needs time to adapt. Receptors change sensitivity. Gene expression shifts. Synaptic strength slowly improves.
Ketamine takes a very different route.
What Is Ketamine and How Did It Enter Psychiatry?
Ketamine’s Medical Origins
Ketamine was originally developed as an anesthetic in the 1960s. It has been widely used in surgery and emergency medicine due to its safety profile and ability to preserve breathing reflexes.
For decades, its psychiatric effects were not the focus. That changed when researchers observed something surprising.
Discovery of Rapid Antidepressant Effects
In the early 2000s, clinical studies revealed that low doses of ketamine could reduce depressive symptoms within hours. This was groundbreaking. For individuals experiencing severe depression or suicidal thoughts, speed matters.
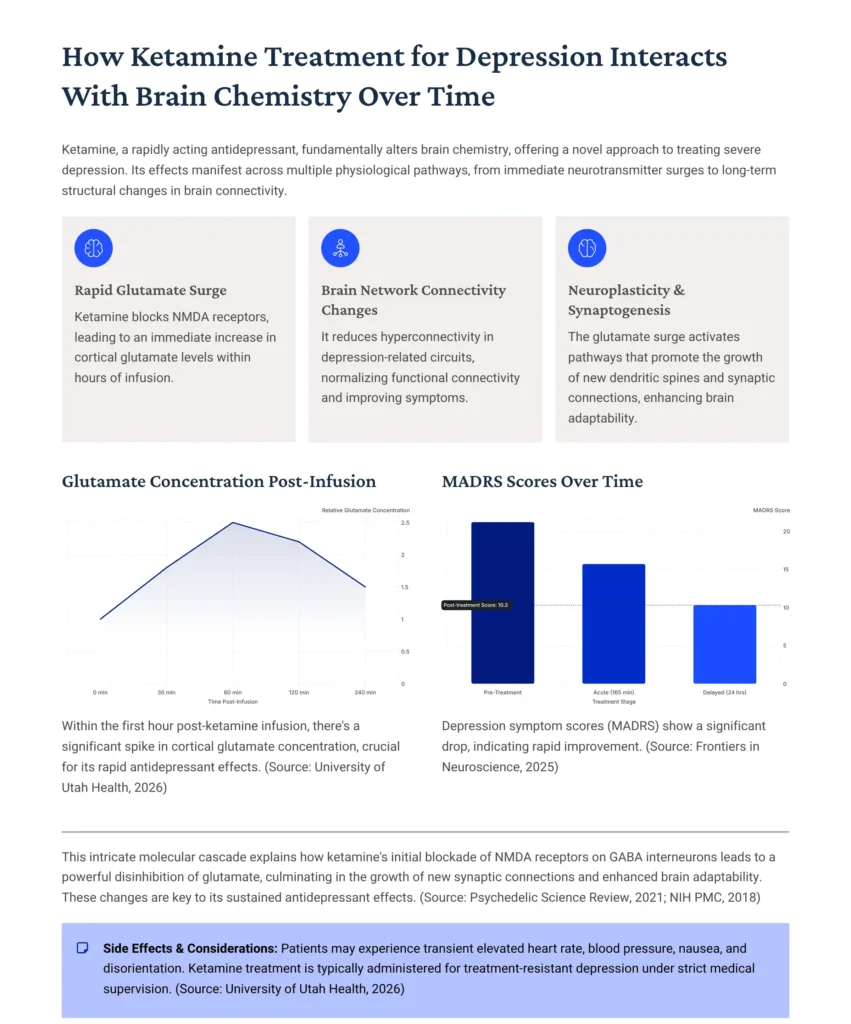
Instead of gradually adjusting serotonin, ketamine appeared to trigger immediate changes in glutamate signaling.
How Ketamine Affects the Brain Differently
NMDA Receptor Blockade Explained
Ketamine primarily works by blocking NMDA receptors. These receptors are part of the glutamate system, the brain’s main excitatory pathway.
Glutamate is like the accelerator pedal of the brain. It drives learning, memory, and adaptation. But under chronic stress, glutamate signaling can become dysregulated.
Glutamate and Synaptic Signaling
When ketamine blocks NMDA receptors on specific inhibitory neurons, it creates a ripple effect. Instead of suppressing activity, this blockade actually leads to a brief surge in glutamate release.
It sounds counterintuitive. Block a receptor to increase activity? But that is exactly what happens.
The Glutamate Surge and AMPA Activation
The temporary glutamate surge activates AMPA receptors. These receptors strengthen synaptic connections and stimulate growth pathways.
This is where the magic begins. Within hours, intracellular signaling pathways such as mTOR activate. These pathways promote protein synthesis necessary for forming new synapses.
In simple terms, ketamine helps the brain build new connections quickly.
Neuroplasticity and Synapse Formation
Brain-Derived Neurotrophic Factor and Growth Pathways
Brain-Derived Neurotrophic Factor, or BDNF, plays a major role in neuroplasticity. Depression is often associated with reduced BDNF levels.
Ketamine increases BDNF release. Imagine fertilizer being poured onto a struggling garden. New growth becomes possible.
Higher BDNF supports dendritic spine formation, which strengthens communication between neurons.
Neuroplasticity is not just a short-term response but a foundational mechanism behind sustained improvement in depressive symptoms. By promoting dendritic spine growth and strengthening synaptic communication, ketamine helps restore functional connectivity in mood-regulating circuits. If you would like a deeper breakdown of how synaptic remodeling supports emotional recovery, explore our detailed discussion on What Role Neuroplasticity Plays in Ketamine Treatment for Depression, where we examine the long-term biological impact of enhanced neural adaptability.
Repairing Stress-Induced Neural Damage
Chronic stress shrinks dendritic branches in the prefrontal cortex. This region is essential for decision making and emotional control.
Ketamine appears to reverse some of this shrinkage. Animal studies show restoration of synaptic density within days. Human imaging studies suggest improved connectivity in mood-related networks.
This is not simply symptom masking. It reflects structural repair.
Short-Term Brain Chemistry Changes After Infusion
Rapid Mood Elevation Mechanisms
Within hours of administration, many individuals report mood improvement. This correlates with increased glutamate transmission and enhanced connectivity between the prefrontal cortex and limbic system.
Functional MRI scans show normalization of overactive circuits linked to rumination.
Reduction in Suicidal Ideation
One of the most studied effects of ketamine is its rapid reduction in suicidal thoughts. The mechanism may involve modulation of the anterior cingulate cortex, an area involved in emotional pain processing.
These short term changes are primarily chemical and electrical in nature.
Medium-Term Adaptations in Neural Circuits
Strengthening Prefrontal Cortex Connectivity
Over days to weeks, repeated treatments can reinforce new synaptic pathways. The prefrontal cortex becomes more capable of regulating emotional responses.
Stronger top down control means fewer overwhelming emotional spikes.
Calming Hyperactive Amygdala Responses
The amygdala is the brain’s alarm center. In depression, it can become overly reactive.
Ketamine appears to reduce amygdala hyperactivity over time, helping to balance emotional reactivity.
Long-Term Brain Chemistry Implications
Sustained Neuroplastic Effects
Long term effects depend on repeated activation of plasticity pathways. Each session can reinforce neural growth, much like exercising a muscle.
The brain gradually stabilizes new patterns of communication.
Maintenance and Brain Stability
Research suggests that maintenance schedules may help preserve synaptic gains. The goal is not constant stimulation but supporting lasting structural resilience.
Some clinics, including Vitality Lounge Med Spa, provide structured treatment protocols. In discussions about Ketamine Treatment for Depression, clinicians often emphasize careful monitoring of long term neurological response.
Comparing Ketamine to SSRIs and Other Antidepressants
Timeline of Neurochemical Changes
SSRIs increase serotonin slowly and indirectly promote neuroplasticity over weeks.
Ketamine triggers rapid glutamate driven plasticity within hours.
Both ultimately aim to improve synaptic health, but their entry points differ significantly.
Different Biological Pathways
SSRIs modulate monoamines. Ketamine modulates glutamate and downstream growth pathways.
This distinction explains why ketamine may work in cases where traditional medications have failed.
Safety and Monitoring Over Time
Cognitive Effects
Short term side effects can include dissociation or perceptual changes. These are linked to acute NMDA receptor blockade.
Most cognitive effects resolve quickly. Long term studies continue to monitor memory and executive function.
Tolerance and Neuroadaptation
Repeated exposure may lead to adaptive changes in receptor sensitivity. Careful scheduling aims to minimize tolerance.
Ongoing monitoring ensures that neurochemical benefits are sustained without unwanted adaptations.
Ongoing Research and Future Directions
Biomarkers and Personalized Psychiatry
Scientists are exploring biomarkers that predict response. Genetic factors, inflammatory markers, and brain imaging patterns may help tailor treatment.
This represents a shift toward precision psychiatry.
Expanding Knowledge of Glutamate-Based Therapies
Ketamine opened the door to a new class of antidepressants targeting glutamate pathways.
Researchers are developing compounds that mimic beneficial effects without dissociative properties.
Vitality Lounge Med Spa Serving the Irishtown Bend Community and Beyond in Cleveland
Vitality Lounge Med Spa is dedicated to serving the diverse needs of the local community of Cleveland, including individuals residing in neighborhood like Irishtown Bend. With its convenient location near landmarks such as the Cuyahoga River and major intersections like Carnagie Ave and Canal Rd (coordinates: Latitude: 41.4930734, Longitude: -81.694086), we offer ketamine treatment for depression services.
Get ketamine treatment for depression services at Irishtown Bend Now
Call Us or Contact Us (216) 202-4770
Navigate from Irishtown Bend to Vitality Lounge Med Spa Now
Depression alters the brain at multiple levels, from neurotransmitter imbalance to structural disconnection. Ketamine interacts with brain chemistry in a layered process.
First, it blocks NMDA receptors. Then it triggers a glutamate surge. Next, it activates growth pathways. Over time, it strengthens synaptic connections and restores healthier communication between key emotional circuits.
Unlike traditional antidepressants that primarily adjust serotonin, ketamine works through glutamate driven neuroplasticity. Its effects unfold across hours, days, and weeks, reshaping neural networks rather than simply boosting chemical levels.
Understanding this timeline helps explain why interest in Ketamine Treatment for Depression Cleveland has grown in clinical discussions. The science continues to evolve, but the core principle remains clear. By promoting rapid and sustained neuroplastic change, ketamine offers a biologically distinct pathway for addressing depressive illness.
FAQs
1. How quickly does ketamine begin changing brain chemistry?
Ketamine begins altering glutamate signaling within hours. Synaptic growth pathways activate rapidly, leading to early mood improvements.
2. Does ketamine permanently change brain structure?
Research suggests it promotes synaptic growth and neuroplasticity. Long term permanence depends on repeated reinforcement and individual brain biology.
3. Why does ketamine work when SSRIs fail?
Ketamine targets glutamate pathways instead of serotonin. This alternative mechanism can help individuals who do not respond to monoamine based medications.
4. Are the brain chemistry changes measurable?
Yes. Imaging studies show changes in connectivity patterns, particularly in the prefrontal cortex and limbic system.
5. Is neuroplasticity the key to long term improvement?
Current evidence strongly suggests that enhanced neuroplasticity is central to ketamine’s sustained antidepressant effects.
